- Home
- About
- Contact
- Blog
- Warframe getamped 2 skins
- Hp laserjet p2055dn driver windows 7 64
- Pano2vr pro license key
- Coreldraw x7 training
- Platinum sx pro multiviewer
- Amores perros free online
- Sketchup version control
- Calcusyn v2
- Watch malayalam movie ennu ninte moideen
- Tetris friends login
- Download sketchup pro 2020
- Wondershare filmora 8-5-3 registration code
- Play game of lust 2
- My rosetta stone activation code only allows 27 characters
- Omnisphere dll locations
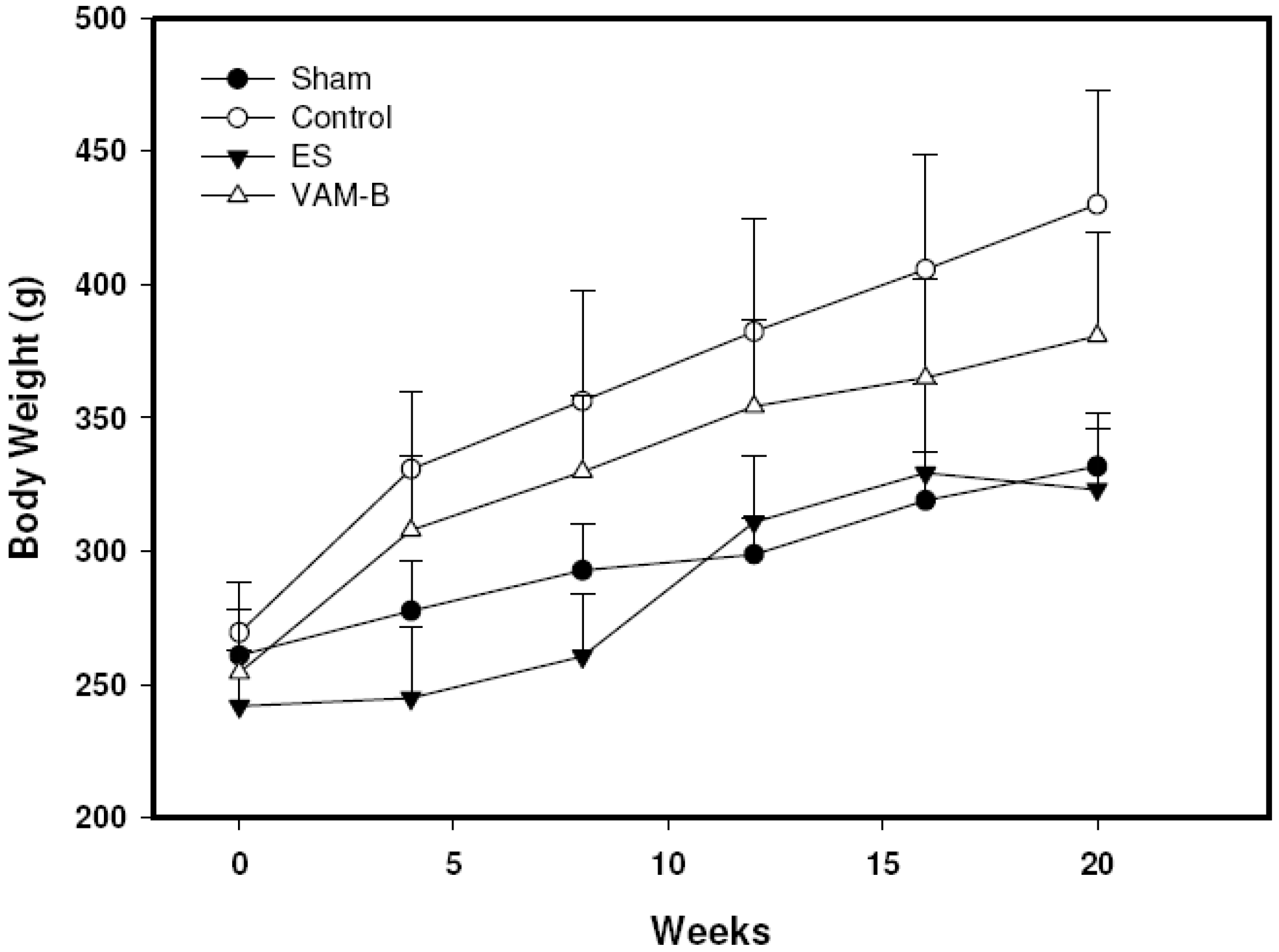
Due to the limited number of chemotherapies available, not least because the development of two novel compounds is doubly challenging, candidates are usually combined with existing treatments. The prioritisation of combinatorial regimes over monotherapy in malaria has meant that the analysis of drug interactions has become an increasingly important part of the drug development pipeline. The novel findings present a potential route to harness the nanomolar antimalarial efficacy of this affordable natural product. We report here the use of the optimised Chou Talalay method to define synergistic antimalarial drug interactivity between emetine dihydrochloride hydrate and atovaquone. The combination was used to further understand the relationship between SYBR Green viability and cytocidal versus cytostatic effects of drugs at higher levels of inhibition. The method, based on the median effect principle proposed by Chou and Talalay, was initially validated for antimalarial application using the known synergistic combination (atovaquone-proguanil).
#Calcusyn v2 software
Here we use isobologram and combination-index data generated by CalcuSyn software analyses (Biosoft v2.1) to define drug interactivity in an objective, automated manner. The lack of reliable and standardised methodology to enable the in vitro definition of synergistic potential for antimalarials is a major drawback. Identification of a synergistic partner drug would present an opportunity for dose-reduction, thus increasing the therapeutic window. Despite its 1000-fold increase in in vitro antimalarial potency (ED 50 47 nM) compared with its anti-amoebic potency (ED 50 26–32 uM), practical use of the compound has been limited by dose-dependent toxicity (emesis and cardiotoxicity). To fast-track antimalarial drug discovery, we have previously employed drug-repositioning to identify the anti-amoebic drug, emetine dihydrochloride hydrate, as a potential candidate for repositioned use against malaria. Unfortunately, antimalarial combination therapy is limited by the depleting repertoire of effective drugs with distinct target pathways. Combination therapies have a range of advantages, including synergism, toxicity reduction, and delaying the onset of resistance acquisition.

Therefore, considering the low toxicity and well-defined clinical characteristics of Art, combination of Sora and Art may present an attractive therapeutic option in the development of clinical trials for HCC treatment.The widespread introduction of artemisinin-based combination therapy has contributed to recent reductions in malaria mortality. Furthermore, combination at a fixed ratio presented mutual enhancement with respect to apoptosis induction and suppression of in vitro liver cancer cell migration. The combination index and dose reduction index were specific to each cell line. The results of the present study revealed that the inhibitory effect of Sora on cell growth was synergistically enhanced by the combination with Art in HepG2 and Huh7 cells. Additionally, a cell migration assay was conducted using Essen ImageLock plates with an IncuCyte Zoom imaging system. Cell apoptosis was evaluated by measurements of the proportion of cells in the sub / phase of the cell cycle, and determination of protein expression levels of cleaved poly ADP ribose polymerase and caspase-9. Drug combination and reduction indices and isobologram plots were used to assess drug interactions. Cell growth inhibition was determined using the CyQUANT proliferation assay in two liver cancer cell lines, HepG2 and Huh7. To determine the potency of this combination, the present study attempted to quantitatively measure the dose-effect relationship of each drug alone and in combination in liver cancer cells in vitro using Calcusyn software. Abstract : An antimalarial medication, artesunate (Art), has exhibited promising anticancer effects with excellent tolerability in various types of cancer, suggesting that it has the potential to be used in combination with sorafenib (Sora) in hepatocellular carcinoma (HCC) treatment.
